
The shape generally resembles a see-saw and this shape is due to the repulsion in bonding and lone pairs of electrons. The molecular geometry of SF4 according to its molecular formula and hybridization is trigonal bipyramidal. This causes an uneven charge distribution (separete centers of -Īnd + charges) and the molecule becomes polar.Trending Questions How big was Pillotex before it went bankrupt? To circle around an object in space? Why did they downgrade Pluto? What is attacking earth and blowing up structures? How far away is dorset? Do mainstream astronomers recognize the existence of Nibiru? Why does a probe need scientific instruments? How long does nightfall-twilight last? How can you tell how far away a strom is? How many cultures or civilizations use equinoxes or solstices in their life? What is meteorite bombardment? How do scienctists determine how long meteorites have been in space? Which military communications satellite is the most advanced to date and consist of 5 satellites in geosynchronous orbits? How do you treat a Globular Maxillary cyst? What are three reasons an astronomers work is important? Why do we sometimes see only part of the near side of the moon? How did continuous bombardment of meteorites and asteroids affect the temperature and size of earth? What planet in our solar system day is more than half as long as its year? What type of elements are usually found in meteorites? For most of history Euerepeans belived that the sun plantes and stThis began to change in the 1500s when a polish astronomer offered his theory that earth revolves around the sun. Also, the compound is polar in nature as the dipole moment is not zero for SF4. Shape of the molecule becomes angular (bent) like water. These pairs and bonding electron-pairs repel each other and the 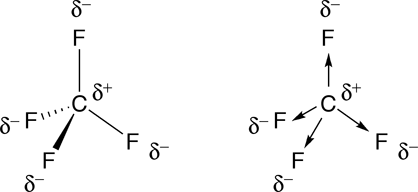
This approach gives no information about the actual arrangement of atoms in space, however. The Lewis electron-pair approach can be used to predict the number and types of bonds between the atoms in a substance, and it indicates which atoms have lone pairs of electrons. But, because of the two nonbondingĮlectron-pairs in the valence shell of O atom, To predict whether a molecule has a dipole moment. Negative charges would coincide with each other, and the molecule If the molecule were linear, the centers of the positive and 
Also, sulfur tetrafluoride has a net dipole moment. 1 Debye when 3.336 × 1030 Coulomb of charge is separated by 1 meter. SF4 Which of the following molecule has a polar bonds but is non SF4 Molecular Geometry, Lewis. unit of dipole moment is Cm (Coulomb meters) or D (Debye). (This is the single compound in which O has Mathematically, the dipole moment of a bond is expressed as: q r Here, q the absolute charge separation between the atoms involved in the bond r distance between the atoms involved in the bond The S.I. hence, the dipole moment will direct towards the fluorine atom. Sides of the molecules become partially negative and oxygen become The fluorine is more electronegative than the xenon atom. Therefore, for each O atom, 2 F atoms are needed to completeįluorine is the most electronegative element, and attracts theīonding electrons to itself more than O. Oxygen has 6 valence electrons and normally can form 2 single orġ double bond (excluding the cases of its coordinate covalentįluorine has 7 valence and it can form only one single bond. Which of the following would have a permanent dipole moment (1) SiF. 
In the Periodic Table, oxygen is in Group VIA and fluorine is a SO3 is trigonal planar so that the individual dipoles on the S-O bonds cancel and the molecule has no dipole moment.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
